A new study on the effectiveness of treatment for spinal cord injuries indicates that a combination therapy, including epidural electrical stimulation (EES) as well as neural stem cells and umbilical cord stem cells, could offer better results than any one treatment on its own.
What is spinal cord injury?
Spinal cord injury is a debilitating, disabling neurological condition resulting from damage to the spinal cord, or to the nerves at the end of the spinal canal. This is most frequently caused by traumatic occurrences such as vehicle accidents, falls, sport injuries or violence, but may also be caused by tumours, infections or degenerative conditions.
Spinal cord injury causes partial or complete loss of sensation and function below the level of the injury, commonly resulting in paralysis (paraplegia or quadriplegia), loss of bladder and bowel control, and breathing issues.[1]
What therapies are currently available?
At present, there are no known therapies that would reverse the initial injury and return an injured spinal cord to full function.[2] Current treatments for spinal cord injury focus on limiting what is called the secondary injury cascade, ideally preventing further damage and thus further loss of feeling and motor function.[3] These methods include surgery, medication, physical therapy and rehabilitation, and assistive devices such as braces or wheelchairs.
What is epidural electrical stimulation for spinal cord injury?
In epidural electrical stimulation, an array of electrodes is implanted along the spinal cord through a surgical procedure called a laminectomy. The stimulation provided by the electrical pulses generated by these electrodes could help in the recovery of functionality following spinal cord injury, improving patients’ ability to walk and stand as well as aiding with bladder and bowel control.[3] [4]
How could stem cells help?
By leveraging the regenerative properties of stem cells, it is hoped that a stem cell treatment could repair and regenerate damaged spinal cord tissue.
This could mean protecting what neurons remain intact, repairing the protective myelin sheath on damaged ones, thus restoring their ability to conduct nerve signals, and replacing lost ones. Stem cells also have the ability to modulate the body’s immune response, and could reduce inflammation and mitigate the secondary damage that follows the initial injury.[5]
There are currently several clinical trials studying the application of stem cells for spinal cord injury. Although more research is needed, results so far are promising, including the high-profile case study of a man who has regained the ability to walk.[6]
What has the new study found?
Researchers at Xi’an Jiaotong University, China, set out to test the effectiveness of a therapy combining both epidural electrical stimulation and stem cell injections, using a mouse model of spinal cord injury.[7]
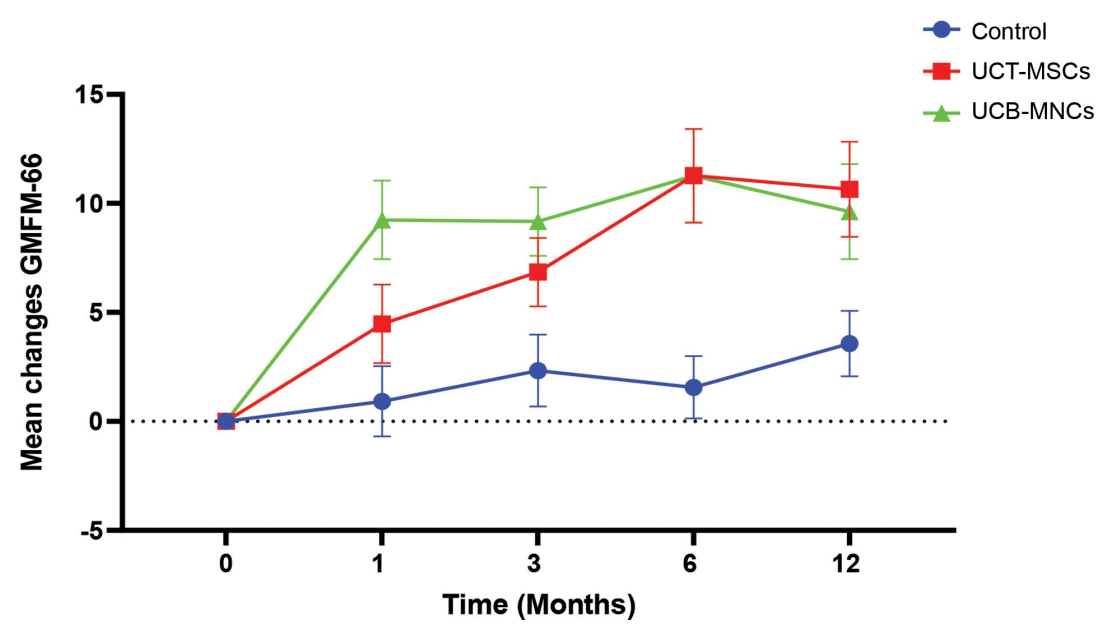
The study involved four different groups of mice: a group which was treated with EES alone, a group which was treated with a mix of mouse neural stem cells (NSCs) as well as human umbilical cord mesenchymal stem cells (hUCMSCs), a group which received both treatments and a control group in which the spinal cord injury was left untreated. The mice in all groups were monitored and assessed for a period of two months.
Following the injury, all mice had complete loss of function in their hind limbs; a week post-injury, mice in the treatment groups underwent their respective treatments. At the end of the monitoring period, mice in the control group were still unable to support themselves on their hind limbs. Conversely, some mice in the EES group were able to achieve paw standing; mice in the hUCMSC group also achieved this milestone, in a more frequent and sustained manner. Mice in the combined treatment group not only achieved paw standing, but also showed improved motor coordination. Swimming and gait analysis tests corroborated these findings, with the hUCMSC group doing better than the EES group, both doing better than the control and the combined treatment group doing best of all.
What’s next?
Both EES and stem cell transplants are currently the subject of clinical trials to test their effectiveness in the treatment of spinal cord injury. As the new study shows, it is entirely possible that the best treatment will be a combination of both, but more research is required on each individual treatment before the combination therapy can be tested in humans.
What is undeniable is that the number of studies and clinical trials examining the regenerative power of stem cells and their potential for treating currently incurable diseases and injuries continues to grow.
To find out more about what stem cells could do, and how you could preserve a rich source of them for your baby so that they could gain access to future regenerative treatments, fill in the form below to request your free welcome pack.
References
[1] University Hospitals Sussex NHS Foundation Trust. (2023). Spinal cord injury. https://www.uhsussex.nhs.uk/sussex-trauma-network/rehabilitation/conditions/spinal-cord-injury/
[2] NIH (2016). What are the treatments for spinal cord injury (SCI)? https://www.nichd.nih.gov/health/topics/spinalinjury/conditioninfo/treatments
[3] Dorrian, R.M., Berryman, C.F., Lauto, A. and Leonard, A.V. (2023). Electrical stimulation for the treatment of spinal cord injuries: A review of the cellular and molecular mechanisms that drive functional improvements. Frontiers in Cellular Neuroscience, 17. doi:https://doi.org/10.3389/fncel.2023.1095259
[4] Royal National Orthopaedic Hospital. (2024). New research offers quality of life hope for many paralysed after spinal cord injuries. https://www.rnoh.nhs.uk/news/new-research-offers-quality-life-hope-many-paralysed-after-spinal-cord-injuries
[5] Zeng, C.-W. (2023). Advancing Spinal Cord Injury Treatment through Stem Cell Therapy: A Comprehensive Review of Cell Types, Challenges, and Emerging Technologies in Regenerative Medicine. International Journal of Molecular Sciences, 24(18), p.14349. doi:https://doi.org/10.3390/ijms241814349
[6] Stem Cells Help Injured Surfer to Walk Again.
[7] Mu, Z., Qin, J., Zhou, X. et al. (2024.) Synergistic effects of human umbilical cord mesenchymal stem cells/neural stem cells and epidural electrical stimulation on spinal cord injury rehabilitation. Sci Rep 14, 26090. doi:https://doi.org/10.1038/s41598-024-75754-x